ExITox-II
Explain Inhalation Toxicity-2
Animal-free mechanism-based toxicity testing – predict toxicity after repeated dose inhalation exposure by using a read across approach.


A collaborative project funded by the German Federal Ministry of Education and Research (BMBF) in the funding program
“e:ToP – Innovative Toxikologie zur Reduzierung von Tierversuchen”
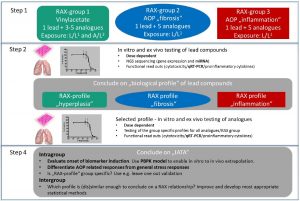
ExITox-2 integrated four new key aspects into the IATA:
- The uptake of compounds after inhalation exposure were estimated by physiological pharmacokinetic modeling and QSAR models.
- The onset of gene changes and miRNA regulation, by dose dependent testing, were investigated to better differentiate between group-specific changes and general stress responses related to high dosing.
- We generated and used master pathways per group to differentiate AOP related changes from general cellular stress responses.
- An IATA was developed, which allows to predict the toxicity of an untested compound in a read across context based on the generated data.