Chemo-Informatics
Compute biological activity for a compound library
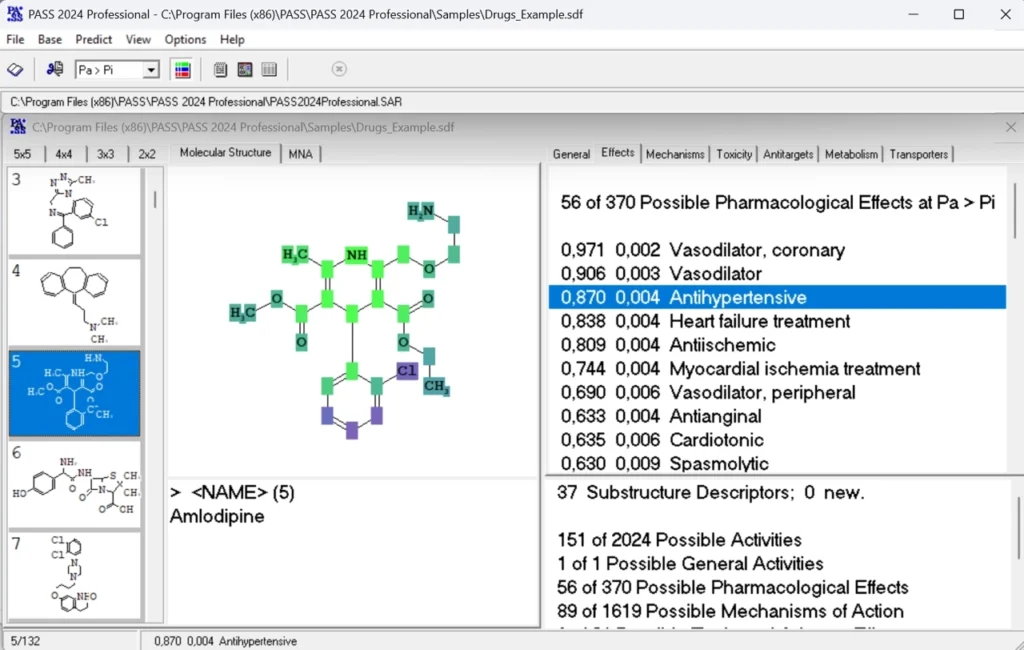
PASS
Compute biological activity for chemical compounds using SAR (structure-activity relationship) approach
- Biggest SAR base – the current release, PASS 2024, comes with the SAR base trained on over 1,480,000 compounds with known biological activities (over 6,000,000 structure-activity pairs)
- Wide spectrum of biological activities — over 9,200 terms (pharmacotherapeutic effects, biochemical mechanisms, toxicity, metabolic effect, gene regulation) can be predicted, and the recommended activity list presents 2,024 activity types.
- High accuracy — average accuracy of predictions exceedes 0.93 for all predictable activities, and is over 0.97 for the recommended activities.
- Drug-Likeness — a new special predictable term, “Drug-Likeness” has been added to the SAR Base of PASS 2024. The term has been trained on over 4,000 compounds.
- Locally installed on your Windows desktop or laptop
- Fast performance – PASS run on a library of hundreds thousands of compounds is done instantly
(depending on the computational power of the system).
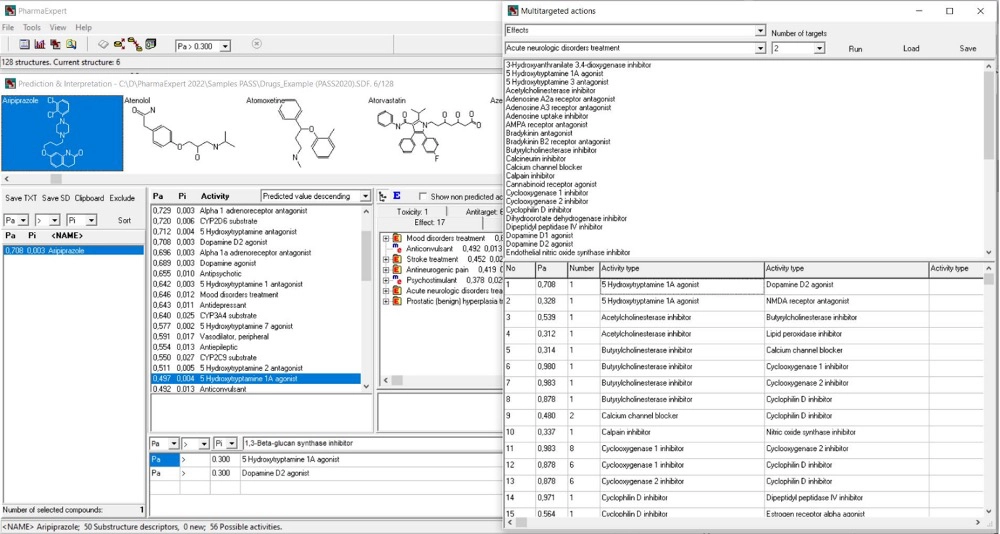
PharmaExpert
Analyze drug mechanisms, effects, and interactions using expert knowledge-based interpretation of PASS results
- Interpretation of PASS prediction results
- Unique manually curated MER base (mechanism – effect relationships) with over 15,700 relationships in the release PharmaExpert 2024
- Selection of compounds with the desired activity spectra predicted by PASS
- Search for multitargeted ligands across mechanisms, pathways, and biological processes
- Comprehensive drug-drug interaction analysis (pharmacokinetic, pharmacodynamic, toxic)