GLI2 in colorectal cancer (CRC)
TFCLass 2.3.3.1.2
GLI1 and GLI2 transcription factors are well documented downstream effectors of the Hedgehog pathway. SRC (SRC-1) is a known coactivator for a big number of different DNA-binding transcription factors.
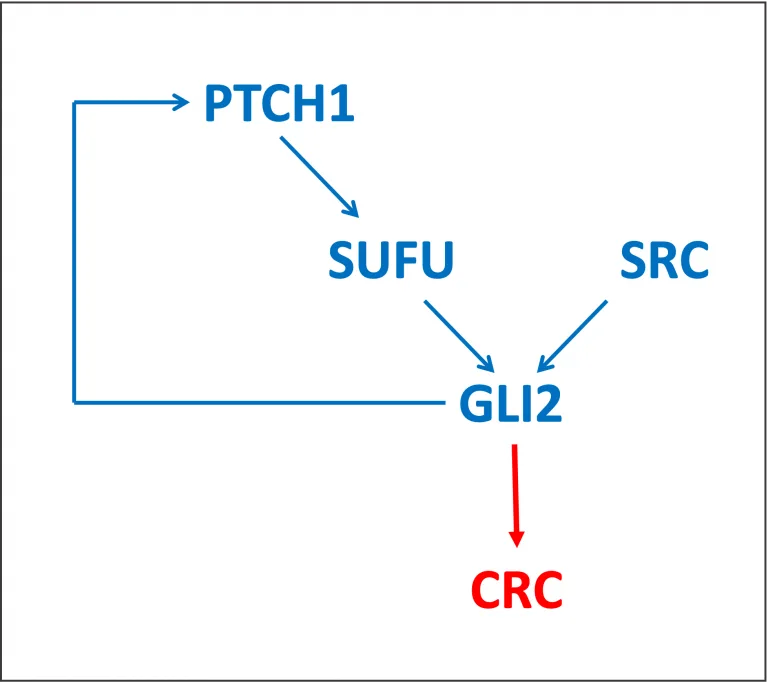
In the recent study SRC was proven as a coactivator for GLI2 in colorectal cancer (CRC). SRC and GLI2 were shown to be highly expressed in the human CRC specimens at both mRNA and protein levels. The direct interactions between GLI2 and SRC were proven and protein domains involved in the interactions were identified. GLI2 interacts with SRC via its DNA-binding zinc finger domains (Guo, P. et al., 2022. Oncogene, 41: 2846–2859). SRC and GLI2 interactions were demonstrated to cooperatively enhance transcriptional activation of GLI2 target genes, BCL2, CCND1 and few others. Gene regulation by SRC and GLI2 promote the Hedgehog signalling leading to cell viability and migration in CRC. This study uncovers some of the molecular mechanisms of the Hedgehog pathway in CRC progression (Guo, P. et al., 2022. Oncogene, 41: 2846–2859).
GLI2, together with 10 other Gli-like factors, belongs to the big class of C2H2 zinc finger factors, especially to a family known as “More than 3 adjacent zinc finger factors” (TFClass link).
GLI2 is known for its association with at least 12 tumor types, the top associated tumors are hepatocellular carcinoma, uterine cervical neoplasms, and basic cell carcinoma (HumanPSD database).
In this publication, GLI2 overexpression in CRC and its role in tumor progression has been studied (Guo, P. et al., 2022. Oncogene, 41: 2846–2859). A disease similarity map for colorectal cancer (CRC) can be viewed here.
Expression of GLI2 protein varies significantly between the organs and tissues investigated. The highest expression level is observed in ovary followed by approximately 2-fold lower expression in vagina, gallbladder, endometrium, cervix, prostate and further down to the neglectable expression in lymphocytes, monocytes, NK-cells and dendritic cells. Expression of GLI2 in normal rectum is quite low, more than 10-times lower as compared to its highest expression level in ovary (HumanPSD database, Human Protein Atlas v20).
High expression levels of GLI2 have been previously demonstrated in several tumor types (HumanPSD database). In this study, a high expression level of GLI2 in CRC was proven (Guo, P. et al., 2022. Oncogene, 41: 2846–2859).
A number of protein interactions of GLI2 are documented (TRANSPATH database). GLI2, similar to GLI1, interacts directly with SUFU, a negative regulator of Hedgehog signalling; with SKI, a repressor of TGF-beta signalling; with KIF7, a microtubule motor protein; with CARM1, a coactivator playing a role in several pathways.
GLI2 is documented to activate transcription of a number of genes, including BCL2 (a known regulator of apoptosis, cell cycle, DNA repair), CCND1 (cyclin D1, cell cycle regulator), CFLAR (apoptosis regulator), FOXA2, FOXE1, MEOX1, MYF5, NANOG (transcription factors).
GLI2 regulates transcription of PTCH1, a hedgehog receptor, as well as GLI1, a downstream transcription factor of the hedgehog signalling (TRANSFAC database).
In TRANSFAC 2.0, there are 10 matrices for GLI2. Among them, there are matrices based on the individual genomic binding sites and on the SELEX experiments. There are two family matrices for Gli-like factors. The core binding motif of GLI2 is TGGGTGGT (TRANSFAC database).
Many more details can be found in the integrated database TRANSFAC + TRANSPATH + HumanPSD, in the Locus Report for human GLI2:
Open this report as a PDF file.
Guo, P. et al., 2022. Nuclear receptor coactivator SRC-1 promotes colorectal cancer progression through enhancing GLI2-mediated Hedgehog signaling. Oncogene 41, 2846–2859. PMID: 35418691
This page was published and last revised on 16.09.2022
Examples of some other transcription factors in cancer can be found here.
