Canonical vs. Non-Canonical Signaling: TNF Superfamily Members Activate Non-Canonical NF-κB Signaling
When you think of cell signaling, it’s tempting to imagine a single, straight road—but NF-κB runs two.
The canonical pathway fires fast to tackle stress and infection, while the non-canonical route, driven by NIK, works more slowly and selectively, shaping immune and developmental responses.
TNF superfamily members are the keys that unlock this second route.Read on, and you’ll see exactly how these pathways differ, why the non-canonical switch matters, and what it reveals about p50:RelA vs. p52:RelB dynamics, disease mechanisms, and even potential therapies.
Difference between Canonical and Non-Canonical NF-kappB pathways
Ligands and Receptors
Canonical NF-κB activation can be initiated by diverse pro‑inflammatory and immune stimuli, including TNF‑α via TNFR, IL‑1β via IL‑1R, LPS via TLRs (especially TLR4), dsRNA via TLR3, antigen receptor signaling through TCRs in T cells and BCRs in B cells, and EDA‑A2 via XEDAR, among others.
In contrast, non‑canonical NF‑κB activation is triggered by a more restricted set of TNF superfamily ligands, such as LTA:LTB and LIGHT via LT‑βR, CD40L via CD40, CD70 via CD27, CD30L via CD30, RANKL via RANK, and THANK/BAFF via BAFFR.
Different cellular surface receptors.
Canonical pathway might be initiated by the following ligands via their receptors:
as well as some other ligands and their receptors.
Non-canonical pathway might be initiated by the following ligands via their receptors:
IKK Complex and Activation Mechanisms
Once receptors are engaged, the intracellular signaling components diverge significantly between the two pathways.
A key distinction is the composition and role of the IKK (IκB kinase) complex and how NF-κB is activated:
Canonical pathway signaling:
The canonical pathway relies on the classical IKK complex and the phosphorylation-induced degradation of an inhibitor protein (IκB). The IKK complex in canonical signaling is a heterotrimer consisting of two catalytic subunits IKKα and IKKβ, and a regulatory subunit IKKγ (also known as NEMO, NF-κB Essential Modulator).
Upon pathway activation, upstream kinases (e.g. TAK1) activate this IKK complex. The active IKK (particularly IKKβ) phosphorylates the inhibitor IκBα (one of the IκB family proteins that bind NF-κB dimers in the cytosol).
Phosphorylation of IκBα marks it for ubiquitination and proteasomal degradation. As IκBα is degraded, it releases the NF-κB dimer. The released NF-κB (most often a p50:RelA heterodimer in canonical signaling) then translocates into the nucleus to regulate target genes.
This process is relatively rapid, providing an immediate NF-κB response to stress or inflammatory signals.
Non-canonical pathway signaling:
The non-canonical pathway does not use the IKKβ/NEMO-containing complex, and it does not primarily rely on IκBα degradation.
Instead, it uses a distinct kinase, NIK (NF-κB–inducing kinase, MAP3K14), to activate a unique IKK complex.
Here, IKK is a homodimer of IKKα subunits (IKKα:IKKα), with no IKKβ or NEMO present.
Under resting conditions, NIK is kept at very low levels through continuous degradation: NIK forms a complex with adaptor proteins TRAF2, TRAF3 (and in some cases TRAF6) and cellular inhibitor of apoptosis proteins cIAP1/2, which ubiquitinate NIK and target it for proteasomal destruction.
Upon ligand binding to TNF superfamily receptors (e.g. BAFFR, CD40, LT-βR), those adaptors (TRAF2/3 and cIAPs) are recruited to the receptor and sequestered away from NIK, preventing NIK’s ubiquitination.
This allows NIK protein to rapidly accumulate. The stabilized NIK
then phosphorylates and activates IKKα homodimers. Active IKKα in turn phosphorylates the NF-κB2 precursor p100 (also known as NFKB2/p100, which is analogous to an IκB-like protein with ankyrin repeats).
Phosphorylation of p100 targets it for partial proteasomal processing: the C-terminal inhibitor domain of p100 is ubiquitinated and degraded, converting p100 into p52.
The newly generated p52 subunit then heterodimerizes with RelB, another NF-κB family member. The p52:RelB dimer translocates to the nucleus to regulate its target genes.
Notably, NIK is the key driver of this entire cascade – all known non-canonical NF-κB inducers work by activating NIK, establishing NIK as the central node in the non-canonical pathway.
This non-canonical route is typically slower and more tightly regulated than the canonical route, often involved in developmental or tissue-specific signaling rather than acute responses.
Differences in the NF-κB subunit composition.
NF-κB family of transcription factors includes several members encoded by different genes.
NFKB1 gene encodes p105 protein, which is processed to NF-κB p50.
NFKB2 gene encodes p100 protein, which is processed to NF-κB-p52.RelA gene encodes p65. Further, there are RelB and RelC genes.
More details about the NF-kappaB family can be found in the Classification of transcription factors.
An active NF-KappaB transcription factor is a heterodimer composed of two subunits.
These dimers can regulate overlapping but distinct gene sets, underpinning different biological outcomes.


Visualization of the NF-κB pathway
Tissue‑, Organ‑, and Cell‑Specific Expression of TNF Superfamily Receptors
The receptors of the non-canonical NF-kappaB pathway are characterized by a tissue-enriched expression, that is their expression is significantly higher in several organs, tissue- or cell-types as compared to other studied tissues. Tissue specificity of the receptors defines a location and physiological function of a particular non-canonical chain leading to p52:RelB activation.
Here, details of tissue-specific expression of several TNF receptor superfamily members are shown.
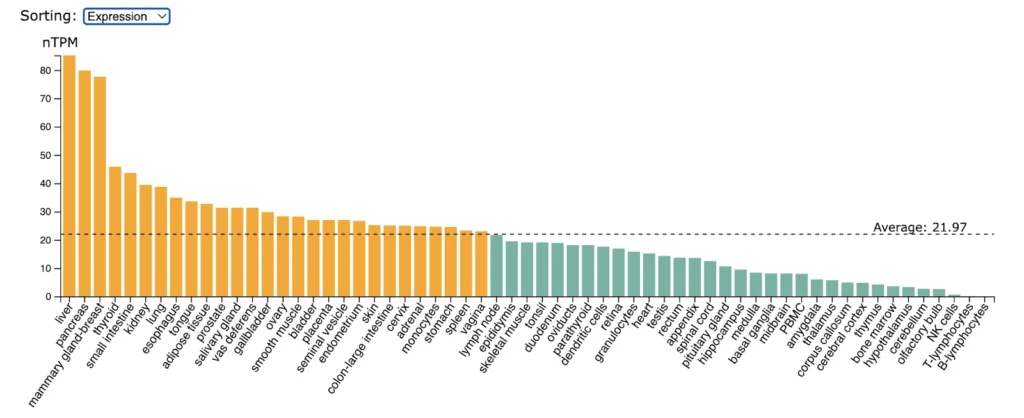
LT-betaR (TNFRSF3) expression is significantly enriched in liver, pancreas, mammary gland.
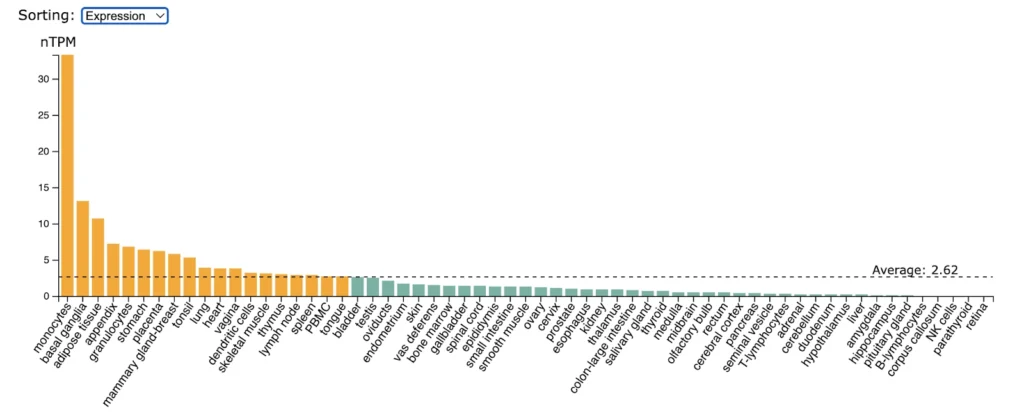
CD30 (TNFRSF8) expression is significantly enriched in monocytes followed by lower but still enriched expression in basal ganglia, adipose tissue, appendix, granulocytes, stomach.
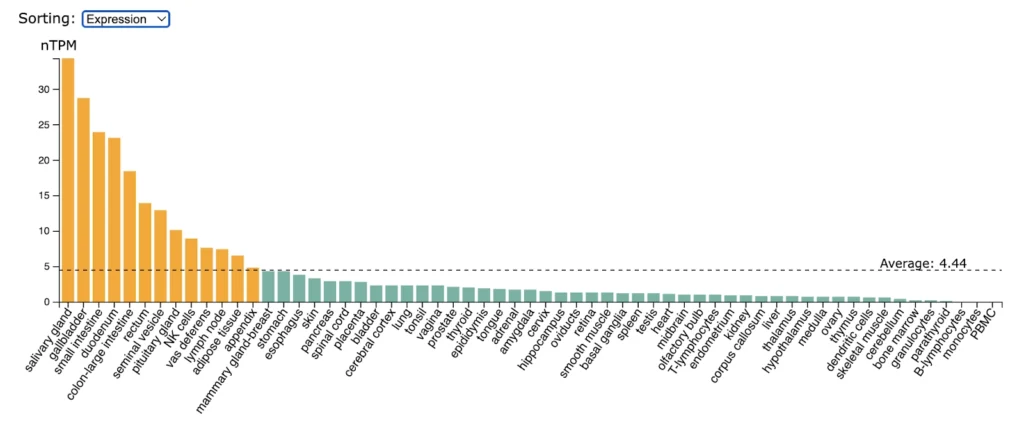
RANK (TNFRSF11A) expression is significantly enriched in salivary gland, gallbladder, small intestine, duodenum, colon, rectum.
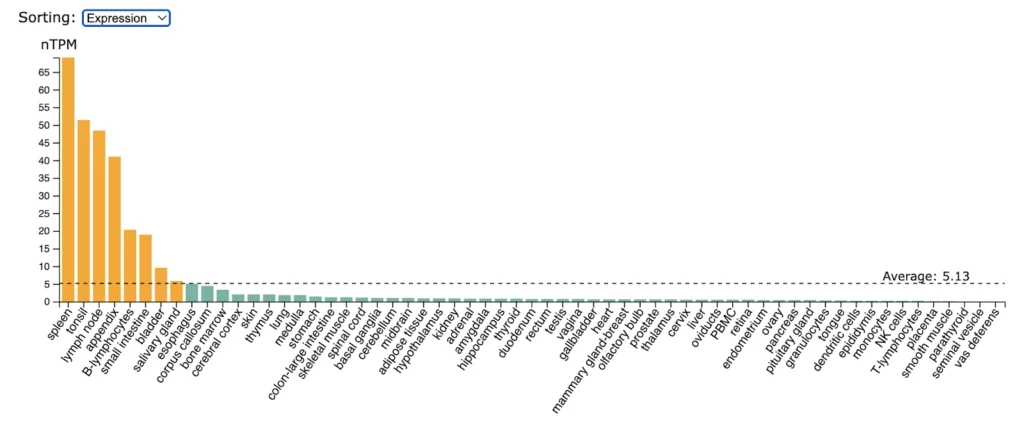
BAFFR (TNFRSF13C) expression is higly enriched in spleen, tonsil, lymp nodes, appendix, B-lymphocytes, small intestine.
Brief summary
In summary, canonical NF-κB signaling is the fast, ubiquitous pathway activated by many receptors and relying on the degradation of IκBα to release p50:RelA dimers, whereas non-canonical NF-κB signaling is a slower, more selective pathway activated by a subset of TNF receptor superfamily (TNFRSF), relying on NIK to drive p100 processing and activation of p52:RelB dimers.
These pathways illustrate how a single transcription factor family can be regulated by multiple upstream routes to tailor the response to specific stimuli and contexts.
While the canonical pathway handles the immediate early response to stress and infection (in virtually any cell), the non-canonical pathway often handles specialized tasks in the immune system and developmental processes.For readers who need deeper, curated detail—including reaction logic, tissue expression and disease involvement of each pathway component as well as pathway/chain visualizations—these details are comprehensively represented in the TRANSFAC Pathways and TRANSFAC Diseases packages.
