HIT-GLIO
Targeting tumor-host interactions in pediatric malignant gliomas to reinvigorate immunity and improve radio- and immunotherapy efficacy
Paediatric high-grade gliomas (pHGGs) are malignant, deadly tumours developing in infants and children. Mutations in histone coding genes disturb epigenetic regulation and in cooperation with other oncogenes promote tumour initiation and progression. pHGGs are fast-growing and diffusive which makes them hard to remove or treat. Surgery (unattainable in midline brain regions) and radiotherapy (RT) remain the only option with transient benefits as median survival is 9-15 months. Those tumours are immunosuppressed, thus immunotherapy is ineffective. The HIT-GLIO projectproposes that knowledge of tumour-host interactions in the tumour microenvironment (TME) would reveal novel targets allowing to find new ways to eradicate mutated tumour cells, improve efficacy of RT, and reinvigorate anti-tumour immunity.
To achieve these ambitious goals within the HIT-GLIO project we have built up the international, multidisciplinary consortium that includes clinicians, medical researchers, biologists, bioinformaticians, and other specialists in the related fields coming from such renown institutions and organizations as Nencki Institute of experimental biology (Poland), Children’s Memorial Health Institute (Poland), Gustave Roussy Institute (France), Centre Nationale de Recherche ScientifiqueCNRS (France), Sanquin (Netherlands), Tel Aviv University (Israel), Istituto Superiore di Sanità(Italy), University of Oxford (UK), McGill University (Canada). We at geneXplain are extremely proud to be a member of such a great consortium.
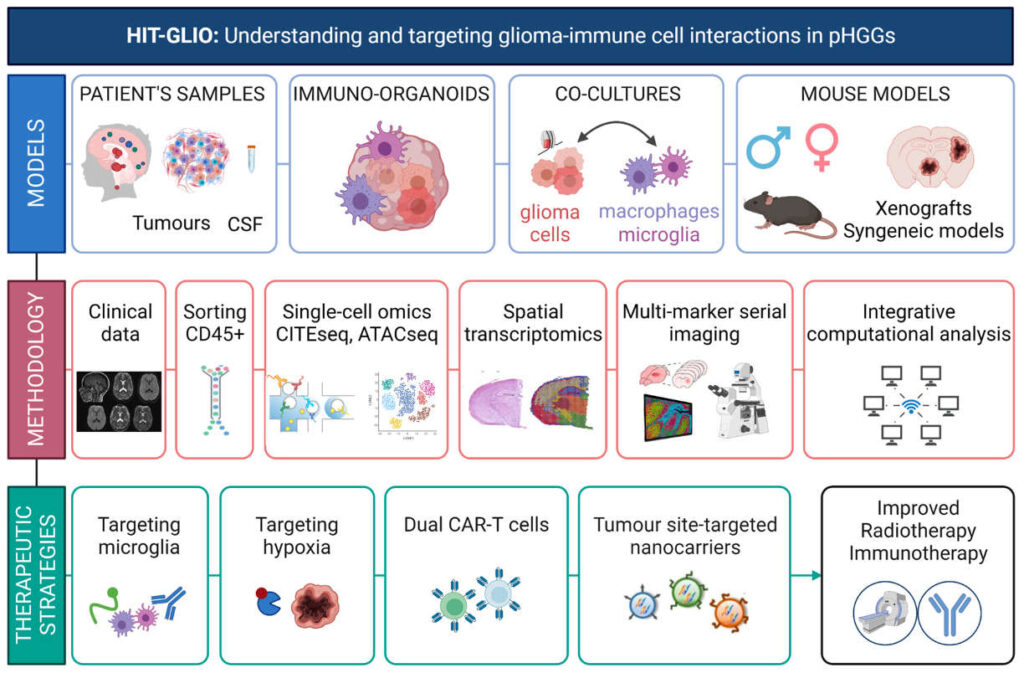
Within the HIT-GLIO project the latest single-cell technologies and multimodal imaging will be usedto characterise tumour-immune cell interactions in a large spectrum of pHGG patient samples. We will create cellular and animal models of increasing complexity: human glioma-microglia co-cultures, DIPG-derived organoids complemented with iPSC-derived immune cells and syngeneic pHGG mouse models which would be platforms for mechanistic studies and drug screening. We will evaluate the hypoxia-inducible epigenetic inhibitors and blockers of tumour-host interactions to improve RT and immunotherapy responses. In addition to that, nanocarriers functionalised to target immune cells and deliver drugs to the brain will be generated. Effective CAR-T cells that together with TME reprogramming and RT would boost anti-tumour immunity will be also developed within the project. We will assess the neurodevelopmental alterations and psychological consequences ofthe disease and foster psychological approaches to increase quality-of-life of patients and caregivers. The expected outcomes of HIT-GLIO project would ultimately pave ways to new clinical trials to improve the way pHGGs are treated.
The summary of models, methodologies, and innovative therapeutic approaches used in the HIT-GLIO project is provided on the image below:
Partners
Nencki Institute of experimental biology (Poland)
Children’s Memorial Health Institute (Poland)
Gustave Roussy Institute (France)
Centre Nationale de Recherche Scientifique CNRS (France)
Sanquin (Netherlands)
geneXplain (Germany)
Tel Aviv University (Israel)
Istituto Superiore di Sanità (Italy)
University of Oxford (UK)
McGill University (Canada)
Coordinator
The project is coordinated by Nencki Institute of experimental biology(Poland)
Contract period
The project duration is 2 years:
01.12.2023-03.11.2025
Contract no.
The project is funded by the Horizon program, Project ID:101136835, within the Call: HORIZON-MISS-2023-CANCER-01 (Research and Innovation actions supporting the implementation of the Mission on Cancer)